Carnot engine, heat engine are some examples of second law of thermodynamics. What is the 2nd law of thermodynamics and give an example? The second law of thermodynamics states that heat can flow spontaneously from a hot object to a cold object heat will not flow spontaneously from a cold object to a hot object. It expresses a fundamental and simple truth about the universe: that disorder, characterised as a quantity known as entropy, always increases. What is the 2nd law of thermodynamics in simple terms? The second law of thermodynamics means hot things always cool unless you do something to stop them. The second law of thermodynamics states that the total entropy of an isolated system (the thermal energy per unit temperature that is unavailable for doing useful work) can never decrease. 1, p. 267 (1988).What does the 2nd law of thermodynamics state? For… In philosophy of physics: Thermodynamics. Smith (eds.) Maximum-Entropy and Bayesian Methods in Science and Engineering vol. The evolution of Carnot's principle, by E.T.Stanford Encyclopedia of Philosophy: " Philosophy of Statistical Mechanics." by Lawrence Sklar.Bristol Philadelphia : Institute of Physics, 2003 Maxwell's demon 2 : entropy, classical and quantum information, computing.
A gentle introduction, a bit less technical than this entry. Reflections on the Motive Power of Fire – and other Papers on the Second Law of Thermodynamics by E.

Sir Arthur Stanley Eddington, The Nature of the Physical World (1927) “ But if your theory is found to be against the second law of thermodynamics I can give you no hope there is nothing for it but to collapse in deepest humiliation. If it is found to be contradicted by observation - well, these experimentalists do bungle things sometimes. If someone points out to you that your pet theory of the universe is in disagreement with Maxwell's equations - then so much the worse for Maxwell's equations.

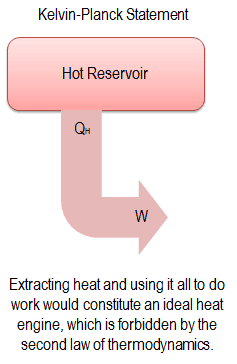
The law that entropy always increases, holds, I think, the supreme position among the laws of Nature. A heat engine is a mechanical device that provides useful work from the difference in temperature of two bodies. Pressure, density and temperature differences in an isolated system, all tend to equalize if given the opportunity density and pressure, but not temperature, are affected by gravity. In a general sense, the second law says that temperature differences between systems in contact with each other tend to even out and that work can be obtained from these non-equilibrium differences, but that loss of thermal energy occurs, when work is done and entropy increases.
